
We apply modern chemical research tools and techniques to develop more sustainable technologies for environmental protection and resource recovery. We are currently developing technologies that employ innovative hydrothermal, photochemical, catalytic, and separations processes to remediate contaminated water, soil, and wastewater biosolids contaminated with per- and polyfluoroalkyl substances (PFAS). We also collaborate with other groups to advance technologies for recovering valuable resources (e.g., fuel, fertilizer) from organic waste streams. Prof. Timothy Strathmann, with more than 25 years of research experience, leads the team’s efforts.
Current research projects include:
- Hydrothermal alkaline treatment of toxic per- and polyfluoroalkyl substances (HALT-PFAS)
- Advancing photochemical treatment technologies for PFAS
- Development of hybrid concentration-destruction treatment trains for PFAS-contaminated water
- New materials and catalysts for hydrothermal waste-to-fuel conversion technologies
- Abiotic mechanisms for transformation of organic contaminants of emerging concern (e.g., pharmaceutically active compounds, PFAS) in soils
- Laboratory and pilot-scale investigation of ion exchange and other adsorption technologies for treatment of groundwater contaminated by PFAS
Hydrothermal alkaline treatment of toxic per- and polyfluoroalkyl substances (HALT-PFAS)
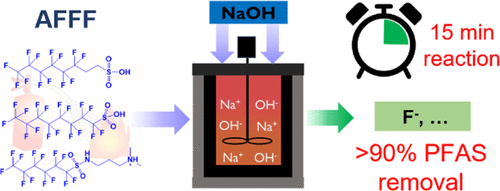 Per- and polyfluoroalkyl substances (PFAS) have been widely used in many commercial and industrial applications since the 1940s, and an increasing number of reports have documented widespread contamination due to inadvertent environmental releases. The extreme persistence, multi-target toxicity, and environmental mobility of PFAS raises serious public health concerns that is leading the federal government and many states to issue proposed regulations that are very stringent. Destruction of PFAS is especially challenging due to the high strength of carbon-fluorine bonds, and most technologies used to treat other classes of organic contaminants are ineffective for treatment of PFAS. Our research group recently introduced an innovative technology for destruction and defluorination of PFAS, hydrothermal alkaline treatment (HALT-PFAS). The HALT-PFAS process combines subcritical hydrothermal water (250-350°C, 5-22 MPa) with low-cost alkali amendments (e.g., NaOH) to promote destruction of PFAS present in water and other high moisture content matrices. To date, our research has demonstrated complete destruction and defluorination of PFAS in groundwater, soil, biosolids, and PFAS-contaminated adsorption media. We were recently issued a patent for the HALT-PFAS technology (US 11,577,111) and are working with industrial partners to commercialize the technology. The HALT-PFAS technology was recently the subject of a CBS News story.
Per- and polyfluoroalkyl substances (PFAS) have been widely used in many commercial and industrial applications since the 1940s, and an increasing number of reports have documented widespread contamination due to inadvertent environmental releases. The extreme persistence, multi-target toxicity, and environmental mobility of PFAS raises serious public health concerns that is leading the federal government and many states to issue proposed regulations that are very stringent. Destruction of PFAS is especially challenging due to the high strength of carbon-fluorine bonds, and most technologies used to treat other classes of organic contaminants are ineffective for treatment of PFAS. Our research group recently introduced an innovative technology for destruction and defluorination of PFAS, hydrothermal alkaline treatment (HALT-PFAS). The HALT-PFAS process combines subcritical hydrothermal water (250-350°C, 5-22 MPa) with low-cost alkali amendments (e.g., NaOH) to promote destruction of PFAS present in water and other high moisture content matrices. To date, our research has demonstrated complete destruction and defluorination of PFAS in groundwater, soil, biosolids, and PFAS-contaminated adsorption media. We were recently issued a patent for the HALT-PFAS technology (US 11,577,111) and are working with industrial partners to commercialize the technology. The HALT-PFAS technology was recently the subject of a CBS News story.
Our group continues to conduct research to advance the HALT-PFAS technology and its application for treatment of a wide range of contaminated liquids and solids, including soils, liquid concentrate byproduct streams, contaminated adsorbent media. A project sponsored by the National Science Foundation’s ERASE-PFAS program, in collaboration with the University of Illinois, aims to apply HALT together with other technologies to treat wastewater biosolids contaminated by PFAS, accomplishing PFAS destruction while simultaneously recovering economically valuable products, including liquid fuels and chemical fertilizers.
Advancing photochemical technologies for remediation of water contaminated with PFAS
 UV light-based advanced reduction processes (UV-ARPs), which generate strongly reducing hydrated electrons (eaq–; -2.9 V), have gained a lot of traction in environmental remediation due to their ability to transform highly oxidized chemicals such as chlorinated solvents and toxic oxyanions at ambient temperatures and pressure. Recent research demonstrates rapid degradation and defluorination of a wide range of PFAS by UV-ARPs. However, wider application of the technology is limited by a lack of understanding of structure-reactivity trends and appropriate models that can be applied to design treatment systems for diverse water sources. Our team has been investigating the underlying mechanisms of hydrated electron reactions with individual PFAS and mixtures of PFAS (e.g., AFFF-impacted groundwater), and developing predictive models for UV-sulfite remediation of PFAS-contaminated groundwater. In collaboration with other groups at Mines, we apply a combination of high resolution mass spectrometry, transient absorption spectroscopy, and density functional theory calculations to study these processes. Recent results have revealed the critical importance of non-target groundwater constituents that act to scavenge hydrated electrons and inhibit reactions with PFAS, including dissolved carbonate species that are ubiquitous in natural water systems. Future work aims to incorporate this information into photochemical models for PFAS treatment that can be validated with groundwater sources collected from different sites where PFAS contamination has been identified.
UV light-based advanced reduction processes (UV-ARPs), which generate strongly reducing hydrated electrons (eaq–; -2.9 V), have gained a lot of traction in environmental remediation due to their ability to transform highly oxidized chemicals such as chlorinated solvents and toxic oxyanions at ambient temperatures and pressure. Recent research demonstrates rapid degradation and defluorination of a wide range of PFAS by UV-ARPs. However, wider application of the technology is limited by a lack of understanding of structure-reactivity trends and appropriate models that can be applied to design treatment systems for diverse water sources. Our team has been investigating the underlying mechanisms of hydrated electron reactions with individual PFAS and mixtures of PFAS (e.g., AFFF-impacted groundwater), and developing predictive models for UV-sulfite remediation of PFAS-contaminated groundwater. In collaboration with other groups at Mines, we apply a combination of high resolution mass spectrometry, transient absorption spectroscopy, and density functional theory calculations to study these processes. Recent results have revealed the critical importance of non-target groundwater constituents that act to scavenge hydrated electrons and inhibit reactions with PFAS, including dissolved carbonate species that are ubiquitous in natural water systems. Future work aims to incorporate this information into photochemical models for PFAS treatment that can be validated with groundwater sources collected from different sites where PFAS contamination has been identified.
Development of hybrid concentration-destruction treatment trains for PFAS-contaminated water
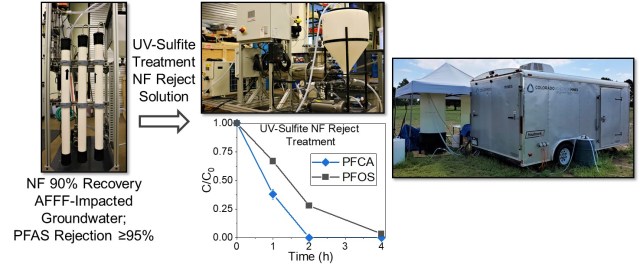 Laboratory investigations and field demonstrations are also underway to study hybrid technologies that combine advanced separations processes (e.g., membrane filtration, adsorption) with destructive processes (e.g., HALT-PFAS) to most efficiently remove and destroy PFAS identified in contaminated water sources. Combining treatment processes can significantly reduce the overall energy requirements for treatment because the volume of PFAS-contamination treated with energy-intensive destruction technologies can be significantly reduced. Working with industrial partners, we are currently applying this strategy to treat PFAS and organic co-contaminants in wastewater from semiconductor fabrication facilities. We also recently completed a field demonstration project treating AFFF-impacted groundwater using an automated treatment train that combined nanofiltration membranes with UV-sulfite treatment of the resulting membrane concentrate stream (see above; see Denver Post story). Other ongoing projects are studying the combination of our HALT-PFAS destruction technology with adsorption, ion exchange, and foam fractionation processes.
Laboratory investigations and field demonstrations are also underway to study hybrid technologies that combine advanced separations processes (e.g., membrane filtration, adsorption) with destructive processes (e.g., HALT-PFAS) to most efficiently remove and destroy PFAS identified in contaminated water sources. Combining treatment processes can significantly reduce the overall energy requirements for treatment because the volume of PFAS-contamination treated with energy-intensive destruction technologies can be significantly reduced. Working with industrial partners, we are currently applying this strategy to treat PFAS and organic co-contaminants in wastewater from semiconductor fabrication facilities. We also recently completed a field demonstration project treating AFFF-impacted groundwater using an automated treatment train that combined nanofiltration membranes with UV-sulfite treatment of the resulting membrane concentrate stream (see above; see Denver Post story). Other ongoing projects are studying the combination of our HALT-PFAS destruction technology with adsorption, ion exchange, and foam fractionation processes.
New materials and catalysts for hydrothermal waste-to-fuel conversion technologies
 Growing population and rising standards of living are driving increases in demand for clean water, and inexpensive energy, materials, and food. At the same time, this produces growing volumes of solid and liquid wastes that public utilities must manage. Current approaches to wastewater treatment expend large amounts of energy to remove organic carbon, nutrients, and pathogens from the water. However, these same “waste” constituents can potentially be recovered as energy or other valuable products while purifying the wastewater. Through integration of biological and catalytic technologies, we can reinvent the wastewater treatment process as a “bio-refinery” that yields clean water as just one product of many. To support these efforts, our research group has been studying the application of catalytic hydrothermal processes for converting waste organic acids (e.g., fatty acids) into hydrocarbon fuels. Catalyst materials can be tailored to promote decarboxylation of these natural waste products into hydrocarbons that are suitable as renewable drop-in replacements for petroleum fuel. Recent work has focused on substituting expensive noble metal catalysts (e.g., platinum, rhodium) with low-cost earth-abundant metals like nickel. We’ve also been investigating strategies to replace the use of external hydrogen gas supplies in these reactions with in situ generation of hydrogen by catalytic reaction of co-occurring organic waste constituents like glycerol.
Growing population and rising standards of living are driving increases in demand for clean water, and inexpensive energy, materials, and food. At the same time, this produces growing volumes of solid and liquid wastes that public utilities must manage. Current approaches to wastewater treatment expend large amounts of energy to remove organic carbon, nutrients, and pathogens from the water. However, these same “waste” constituents can potentially be recovered as energy or other valuable products while purifying the wastewater. Through integration of biological and catalytic technologies, we can reinvent the wastewater treatment process as a “bio-refinery” that yields clean water as just one product of many. To support these efforts, our research group has been studying the application of catalytic hydrothermal processes for converting waste organic acids (e.g., fatty acids) into hydrocarbon fuels. Catalyst materials can be tailored to promote decarboxylation of these natural waste products into hydrocarbons that are suitable as renewable drop-in replacements for petroleum fuel. Recent work has focused on substituting expensive noble metal catalysts (e.g., platinum, rhodium) with low-cost earth-abundant metals like nickel. We’ve also been investigating strategies to replace the use of external hydrogen gas supplies in these reactions with in situ generation of hydrogen by catalytic reaction of co-occurring organic waste constituents like glycerol.
Abiotic mechanisms for transformation of organic contaminants of emerging concern in soils
 Our team is also actively studying naturally occurring abiotic processes and mechanisms that contribute to the transformation of organic contaminants of emerging concern (e.g., pharmaceutically active compounds, flame retardants, PFAS) in soil and groundwater. Growing scarcity of water supplies, particularly in the southwestern United State, is leading more utilities to implement wastewater reclamation projects, often using reclaimed water for irrigation of food crops and turf. Better understanding of the abiotic processes that contribute to degradation of trace organic micropollutants is needed to develop more accurate environmental fate and risk assessment models. Our group is specifically focusing on studying the influence of natural mineral solids and redox-active chemical species on rates and pathways of organic micropollutant degradation. Sensitive and high resolution mass spectrometry methods are being used to monitor a suite of trace organic pollutants and identify major transformation products. Most recently, we are investigating the importance of abiotic mechanisms for the transformation of polyfluoroalkyl substances into more persistent perfluoroalkyl acids like PFOA and PFOS.
Our team is also actively studying naturally occurring abiotic processes and mechanisms that contribute to the transformation of organic contaminants of emerging concern (e.g., pharmaceutically active compounds, flame retardants, PFAS) in soil and groundwater. Growing scarcity of water supplies, particularly in the southwestern United State, is leading more utilities to implement wastewater reclamation projects, often using reclaimed water for irrigation of food crops and turf. Better understanding of the abiotic processes that contribute to degradation of trace organic micropollutants is needed to develop more accurate environmental fate and risk assessment models. Our group is specifically focusing on studying the influence of natural mineral solids and redox-active chemical species on rates and pathways of organic micropollutant degradation. Sensitive and high resolution mass spectrometry methods are being used to monitor a suite of trace organic pollutants and identify major transformation products. Most recently, we are investigating the importance of abiotic mechanisms for the transformation of polyfluoroalkyl substances into more persistent perfluoroalkyl acids like PFOA and PFOS.
Laboratory and pilot-scale investigation of ion exchange and other adsorption technologies for treatment of groundwater contaminated by PFAS
 An alternative to GAC adsorbents, anion exchange resins (AERs) are receiving increased attention for PFAS treatment due to their high selectivity and extended media lifetimes. Our research team has been conducting research on AER treatment of PFAS, both in the laboratory and field settings, to better understand the important factors that govern treatment efficacy. Results of laboratory studies show that adsorption to AERs is highly dependent upon both PFAS chemical structure and resin characteristics. This results from the fact that PFAS adsorb to resins by a combination of electrostatic and hydrophobic interactions. For example, polystyrene-based AERs are much more effective in adsorbing PFAS than polyacrylic-based resins. Field pilot tests confirm trends observed in laboratory bath experiments, including the superiority of resins that incorporate more hydrophobic alkyl chains on the quaternary amine functional groups. Finally, the results of experimental studies are used to inform life cycle assessment and life cycle cost analyses of ion exchange treatment processes in comparison with other technologies. Future work aims to better understand the importance of variable sourcewater chemistry on treatment of PFAS, something that will be critical for wider adoption of ion exchange treatment technologies throughout the nation.
An alternative to GAC adsorbents, anion exchange resins (AERs) are receiving increased attention for PFAS treatment due to their high selectivity and extended media lifetimes. Our research team has been conducting research on AER treatment of PFAS, both in the laboratory and field settings, to better understand the important factors that govern treatment efficacy. Results of laboratory studies show that adsorption to AERs is highly dependent upon both PFAS chemical structure and resin characteristics. This results from the fact that PFAS adsorb to resins by a combination of electrostatic and hydrophobic interactions. For example, polystyrene-based AERs are much more effective in adsorbing PFAS than polyacrylic-based resins. Field pilot tests confirm trends observed in laboratory bath experiments, including the superiority of resins that incorporate more hydrophobic alkyl chains on the quaternary amine functional groups. Finally, the results of experimental studies are used to inform life cycle assessment and life cycle cost analyses of ion exchange treatment processes in comparison with other technologies. Future work aims to better understand the importance of variable sourcewater chemistry on treatment of PFAS, something that will be critical for wider adoption of ion exchange treatment technologies throughout the nation.
